 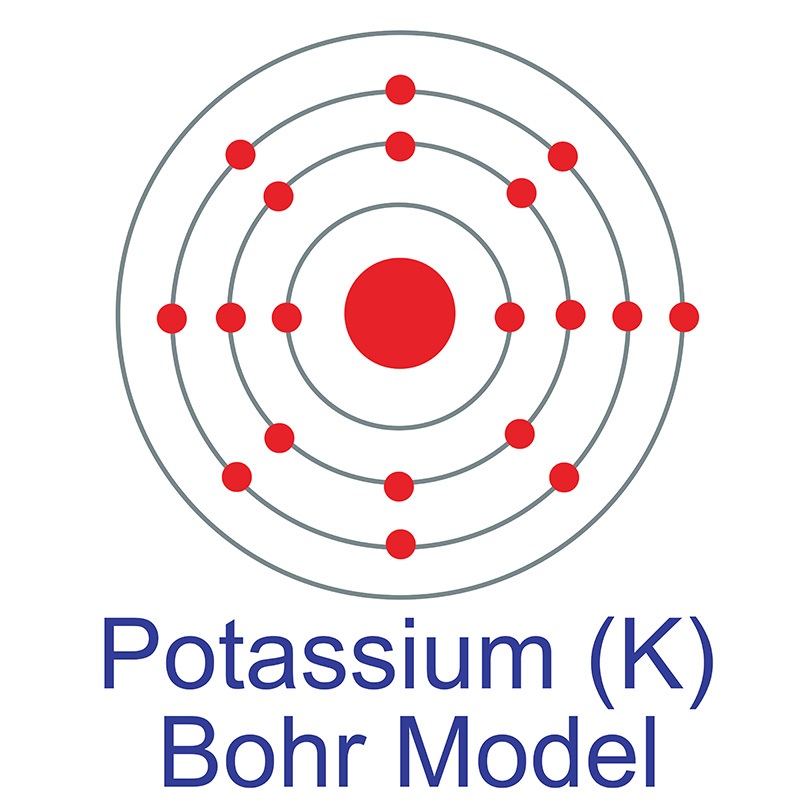 
Both of these alloys produce metals of low vapor pressure and melting points. Electronic configurations model how electrons are arranged in. Potassium atom is Ar 4s1. The ground state abbreviated electronic configuration of Neutral. Because the reaction attains equilibrium quickly, potassium can be removed continuously as a product in order to shift equilibrium to the right and produce even more potassium in its place.Īlloys of potassium include \(NaK\) (Sodium) and \(KLi\) (Lithium). In the modern periodic table, elements are in order of atomic number in periods and groups. Ground State Electronic Configuration of Potassium - neutral Potassium atom Abbreviated electronic configuration of. The reaction is heated in a special device equipped with a furnace, heat-exchanger tubes, a fractionating column, a \(KCl\) feed, a waste removal system, and a vapor condensing system. The isotope shown here is potassium-40, with a nucleus of 21 protons (red) and 19 neutrons (blue). In 1814, the Swedish chemist Berzelius advocated the name kalium for potassium, with the chemical symbol K.\] In 1807, Humphry Davy produced the element via electrolysis: in 1809, Ludwig Wilhelm Gilbert proposed the name Kalium for Davy's "potassium". In 1797, the German chemist Martin Klaproth discovered "potash" in the minerals leucite and lepidolite, and realized that "potash" was not a product of plant growth but actually contained a new element, which he proposed calling kali. The symbol K stems from kali, itself from the root word alkali, which in turn comes from Arabic: القَلْيَه al-qalyah 'plant ashes'. When Humphry Davy first isolated the pure element using electrolysis in 1807, he named it potassium, which he derived from the word potash. Fill orbitals following the model until all electrons have been accounted for. Find the amount of electrons in the atom. 1s is the closest and lowest energy orbital to the nucleus. Example: 1s 2 For writing ground state electron configurations, a few main steps should be followed. Electron configuration of Potassium is Ar 4s1. It reacts violently with water and can even catch fire spontaneously in water. 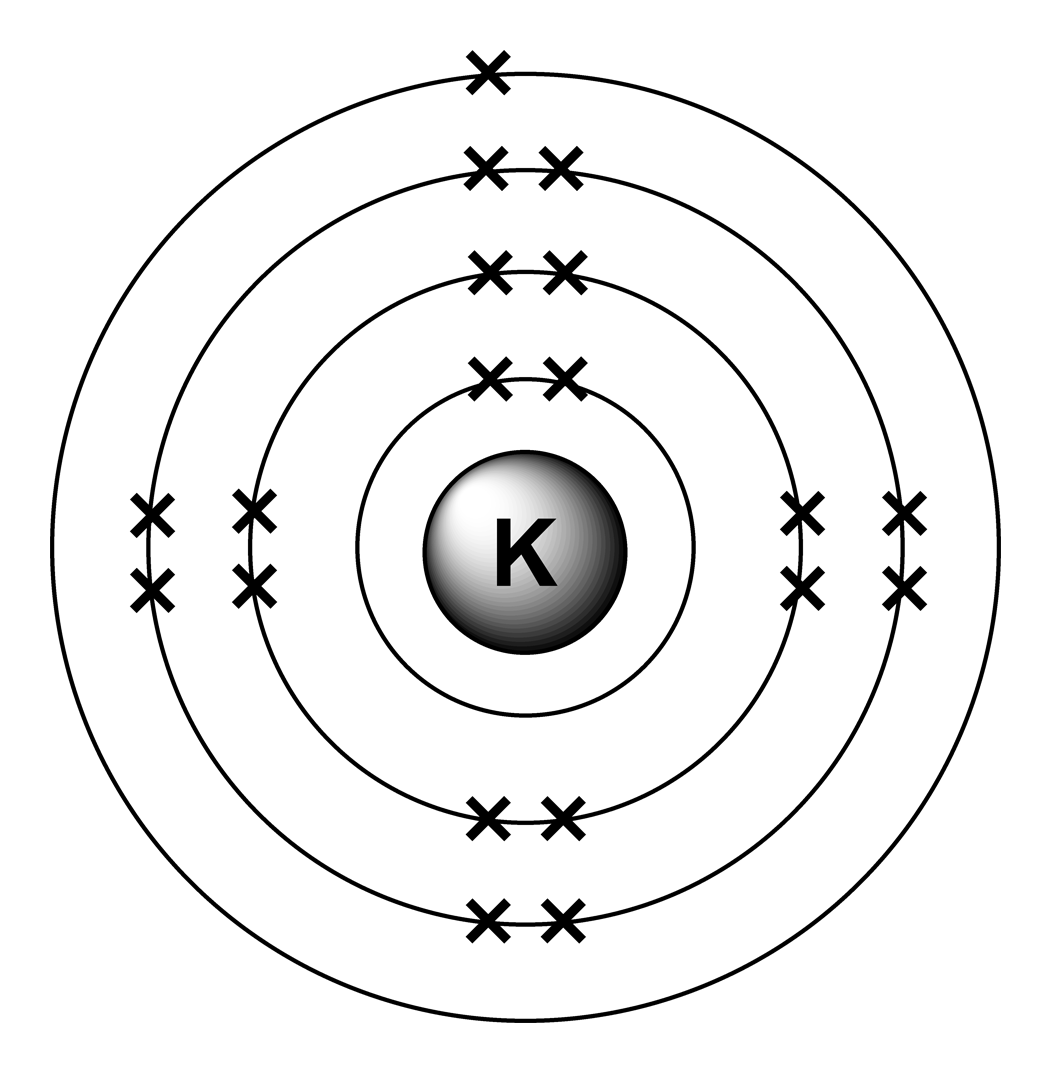
Potassium, the second lightest metal, was discovered by Sir Humphry Davy in 1807. In the periodic table, the elements are listed in order of increasing atomic number Z. Protons/Electrons: 19 Neutrons: 20 Shell structure: 2,8,8,1 Electron configuration: Ar4s1 Oxidation state: 1 Crystal structure: cubic body centered. Which has been discussed in detail above. The number of electrons in each element’s electron shells, particularly the outermost valence shell, is the primary factor in determining its chemical bonding behavior. Then, since the lithium ion has one less electron, remove an electron from. To write the orbital diagram of potassium, you have to write the orbital notation of potassium. 
For example, to find the configuration for the lithium ion (Li), start with neutral lithium (1s☢s¹). Then, add or remove electrons depending on the ions charge. Discovery of potassiumThe English name for the element potassium comes from the word potash, which refers to an early method of extracting various potassium salts: placing in a pot the ash of burnt wood or tree leaves, adding water, heating, and evaporating the solution. To find the electron configuration for an ion, first identify the configuration for the neutral atom. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
